Introduction
The intracellular signaling pathways make possible a
cell surface information transmission , most often from a receptor
cell, to the cell nucleus where gene expression occurs
is going to be modulated through a cascade of intracytoplasmic protein activation.
They are multiple and we will present in this article the most important,
which are particularly involved in cell activation and expression of
pro-inflammatory cytokines that play a critical role in rheumatism
inflammatory. The intracellular signaling pathways described here relate to NF-κB (nuclear factor kappa B), MAP kinase activation pathways
(MAPKs) and JAK / STATs.
The NF-κB signaling pathway
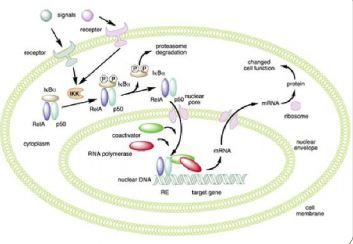
The nuclear factor NF-κB (Figure 1) is a family composed of 5 proteins
fixing DNA and regulating the expression of a large number of genes involved in various biological functions such as immunity,
inflammation, development and apoptosis.
They consist of homodimers and heterodimers which are
sequestered in the absence of activation in the cytoplasm in
combining with inhibitory proteins of NF-κB (IκB). IκB
kinase (IKK) phosphorylates, ubiquitin and degrades IκB, which
results in the release of NF-κB which enters the nucleus where
it activates target genes. Gene transcription mediated by
NF-κB is also regulated by post-transcriptional modifications. Activation of IKK depends on proteins adapters located upstream of the signaling channel, such as TRAF and RIP proteins.
Thus the signaling pathway of NF-estB consists of dimers
NF-κB, IκB proteins, IKK complexes and proteins
intracellular adapters, in particular from the TRAF family
(TNF receptor associated factors).
The activation path MAP kinases (MAPK)
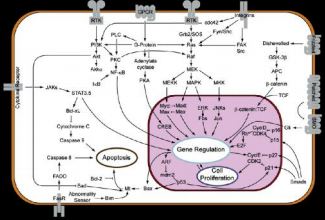
The pathway of MAP kinases (“mitogen activated protein kinases”)
is one of the main routes of transmission of
proliferation signals provided by growth factors such as NGF (“nerve growth factor”). This way involves, after activation of receptors and through adapter proteins, activation of a protein
Ras, at the origin of the cascade of phosphorylation activities: MAP kinase kinase kinase (MKKK or MAP3K), MAP
kinase kinase (MKK, MEKK or MAP2K) and MAP kinase (MAPK).
A MAP3K which is activated by extracellular stimuli
phosphorylates a MAP2K on its serine and threonine residues,
then this MAP2K activates a MAP kinase through the
phosphorylation of its serine and threonine residues. This
last, translocated in the cell nucleus, phosphorylated
then the transcription factors that activate transcription
of all the genes responsible for the replication of
DNA and the initiation of the cell cycle (DNA
polymerases, cyclins, etc.). Thus, MAP kinases are
proteins which have kinase activity which phosphorylates
serine / threonines in response to extracellular stimuli
such as mitogens and which thus regulate activities
various cells such as gene expression, mitosis,
cell differentiation, proliferation and survival and its
corollary, apoptosis or programmed cell death.
Six main groups of MAPKs have been identified in
mammals:
1) Kinases regulated by an extracellular signal (ERK1,
ERK2). ERK1 / 2 kinase pathway activation (also
known as classic MAP kinases) is observed in
response to growth factors or phorbol ester, and
regulate cell proliferation and cell differentiation.
2) C-Jun N-terminal kinases (JNKs). They understand
MAPK8, MAPK9 and MAPK10 and are also known as
name of protein kinases associated with stress (SAPKs). They
in particular activate the pro-inflammatory genes by allowing
the binding of a cJun / c-Fos complex at the promoter level
gene.
3) The p38 isoforms. There are several types: p38-
α (MAPK14), p38-β (MAPK11), p38-γ (MAPK12 or ERK6) and
p38-δ (MAPK13 or SAPK4). Signaling channels p38 and
JNK are activated in response to stress stimuli such as
cytokines, ultraviolet irradiation, shock
thermal or osmotic and are involved in cell differentiation and apoptosis. Activation of MAPK11
results in increased production of TNFα by the macrophage
stimulated by lipopolysaccharide (LPS).
4) ERK5 (MAPK7). It is a discovered kinase
recently which is activated both by factors of
growth and by stress and that participates in proliferation
cellular.
5) ERK3 / 4. ERK3 (MAPK6) and ERK4 (MAPK4) are
Atypical but structurally close MAPKs which have
a SEG pattern in their activation loop and which differ
only at their C-terminus. ERK3 and
ERK4 are essentially cytoplasmic proteins which
attach to and activate MK5 (PRAK, MAPKAP5). ERK3 is
unstable, while ERK4 is relatively stable
6) ERK7 / 8 (MAPK15). This is a discovery MAPK
recently behaving like atypical MAPKs and
has a long C-terminal end similar to ERK3
and ERK4.
The signaling path JAK / STAT
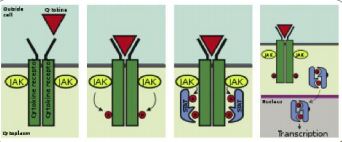
The JAK / STAT signaling pathway participates in the
regulation of cellular response to cytokines and factors
growth. Following activation by a cytokine or a
growth factor, the signaling pathway uses
JAK proteins (Janus kinases) and STATs (Signal transducers
and activators of transcription) to transmit the extracellular signal to the nucleus in which the activated STAT proteins
modulate gene expression.
This signaling channel
plays a critical role in cell proliferation and cell differentiation and apoptosis. She is particularly
important in hematopoiesis. JAKs proteins that have tyrosine kinase activity bind
on certain cytokine receptors. Binding the ligand to
its receiver will activate JAK. The kinetic activity of JAK being
increased will result in increased phosphorylation of
tyrosine residues on the receptor and thus create sites of interaction with proteins which contain SH2 domains
fixing phosphotyrosines. STAT proteins have
SH2 domains capable of fixing these phosphotyrosine residues which are thus recruited on the receptors and are
themselves phosphorylated at the level of their tyrosine residues
by JAKs.
These phosphotyrosines will then serve as a fixing
for SH2 domains of other STATs molecules, thus promoting their dimerization. Thus different STAT proteins can
form homodimers or heterodimers. These dimers
STATs thus activated will translocate to the cell nucleus
and activate transcription of target genes . In addition,
STATs can also be directly phosphorylated at the level
of their tyrosine residues by tyrosine kinases present
at the receptor level (eg EGF receptor or “Epidermal
growth factor “) or by c-src-type tyrosine kinases.
The JAK / STAT signaling channel is regulated in several
levels. Cellular phosphatases can remove
phosphates of cytokine receptors as well as STATS
activated .
SOCS proteins have recently been identified
(“Suppressors of cytokine signaling”) which inhibit the phosphorylation of STATs by fixing and blocking JAKs or while entering into competition with the STATs for the fixing sites
phosphotyrosines at receptors
cytokines. STATS are also negatively regulated by
activated STAT inhibitor proteins (PIAS) that act
at the nucleus.
PIAS1 and PIAS3 thus inhibit transcriptional activation mediated by STAT1 and STAT3 by fixing on them or blocking their access to the sequences that they recognize.
Conclusion
The intracellular signaling pathways, in particular of NF-κB,
MAPKs and JAK / STATs play a critical role in
numerous cellular functions such as proliferation,
differentiation, apoptosis, inflammation and immunity. Their
molecular understanding has come a long way since
a few years and helps explain the mechanisms at the
base of number of pathologies. We can also consider
that in the near future they may be the target of
new therapeutics, especially in the areas of
inflammation, cancer and infectious diseases.
